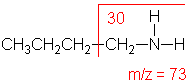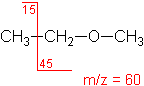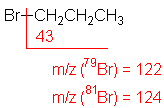Introduction to Mass Spectrometry
-- Examples
Introduction to Mass Spectrometry
-- Examples
Following are examples of compounds listed
by functional group, which demonstrate patterns which can be seen in mass
spectra of compounds ionized by electron impact ionization. These
examples do not provide information about the fragmentation mechanisms
that cause these patterns. Additional information can be found in
mass spectrometry reference books.
Scroll through
this page, or use this list to move to select functional groups (QUIZ!
links you to a tutorial quiz for that functional group):
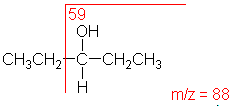
Alcohol:
An alcohol's molecular ion is small or non-existent.
Cleavage of the C-C bond next to the oxygen usually occurs. A loss
of H2O may occur as in the spectra below.
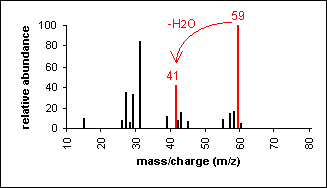
3-Pentanol
C5H12O
MW = 88.15
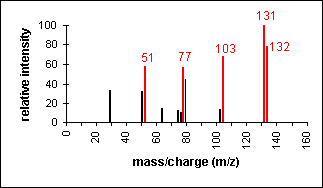
Aldehyde
Cleavage of bonds next to the carboxyl group
results in the loss of hydrogen (molecular ion less 1) or the loss of CHO
(molecular ion less 29).
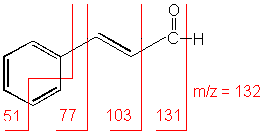
3-Phenyl-2-propenal
C9H8O
MW = 132.16
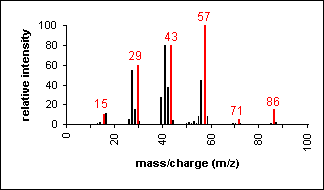
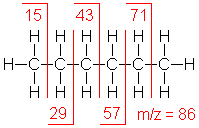
Alkane
Molecular ion peaks are present, possibly
with low intensity. The fragmentation pattern contains clusters of
peaks 14 mass units apart (which represent loss of (CH2)nCH3).
Amide
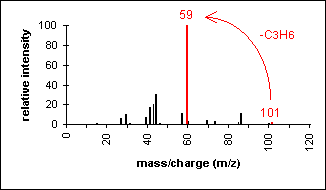
Primary amides show a base peak due to the
McLafferty rearrangement.
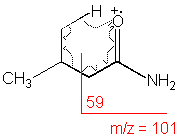
3-Methylbutyramide
C5H11NO
MW = 101.15
Amine
Molecular ion peak is an odd number.
Alpha-cleavage dominates aliphatic amines.
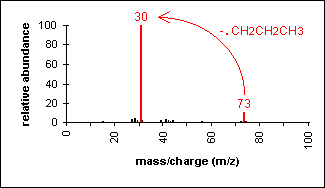
n-Butylamine
C4H11N
MW = 73.13
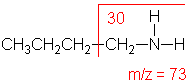
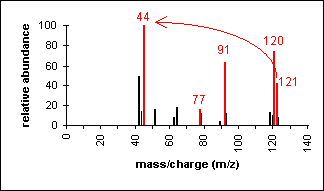
Another example is a secondary amine shown
below. Again, the molecular ion peak is an odd number. The
base peak is from the C-C cleavage adjacent to the C-N bond.
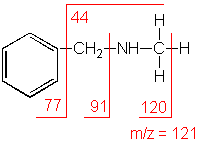
n-Methylbenzylamine
C8H11N
MW = 121.18
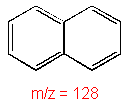
Aromatic
Molecular ion peaks are strong due to the
stable structure.
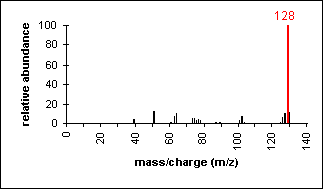
Naphthalene
C10H8
MW = 128.17
Carboxylic
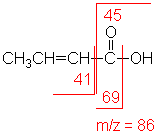
Acid
In short chain acids, peaks due to the loss
of OH (molecular ion less 17) and COOH (molecular ion less 45) are prominent
due to cleavage of bonds next to C=O.
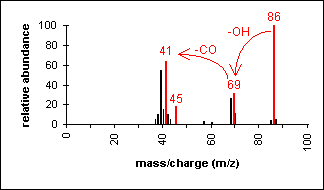
2-Butenoic acid
C4H6O2
MW = 86.09
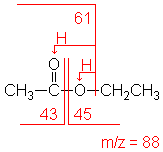
Ester
Fragments appear due to bond cleavage next
to C=O (alkoxy group loss, -OR) and hydrogen rearrangements.
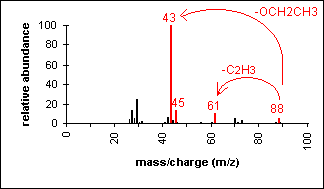
Ethyl acetate
C4H8O2
MW = 88.11
Ether
Fragmentation tends to occur alpha to the oxygen atom (C-C bond next to the oxygen).
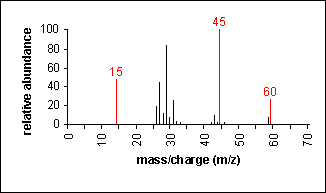
Ethyl methyl ether
C3H8O
MW = 60.10
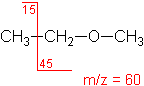
Halide
The presence of chlorine or bromine atoms
is usually recognizable from isotopic peaks.
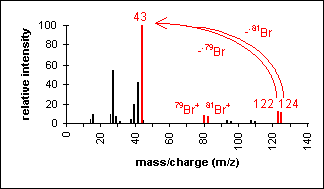
1-Bromopropane
C3H7Br
MW = 123.00
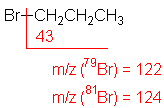
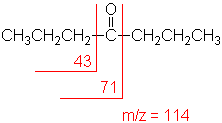
Ketone
Major fragmentation peaks result from cleavage
of the C-C bonds adjacent to the carbonyl.
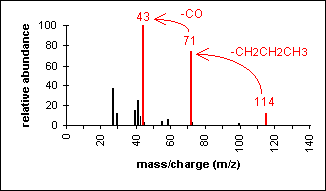
4-Heptanone
C7H14O
MW = 114.19
Return to TOP OF PAGE
Return to CONTENTS
If you have comments or suggestions,
email me at breci@u.arizona.edu